Difference between revisions of "Gnaiger 2018 MiPschool Tromso A1"
| (16 intermediate revisions by 2 users not shown) | |||
| Line 1: | Line 1: | ||
{{Abstract | {{Abstract | ||
|title=[[File:Gnaiger | |title=[[File:Erich Gnaiger.jpg|left|90px|Erich Gnaiger]] Mitochondrial states and rates: 1. Electron transfer pathways and respiratory control. 2. Coupling control. | ||
|info=[[MitoEAGLE]] | |info=[[MitoEAGLE]] | ||
|authors=Gnaiger E | |authors=Gnaiger E | ||
| Line 6: | Line 6: | ||
|event=MiPschool Tromso-Bergen 2018 | |event=MiPschool Tromso-Bergen 2018 | ||
|abstract=[[Image:MITOEAGLE-logo.jpg|left|100px|link=http://www.mitoglobal.org/index.php/MITOEAGLE|COST Action MitoEAGLE]] | |abstract=[[Image:MITOEAGLE-logo.jpg|left|100px|link=http://www.mitoglobal.org/index.php/MITOEAGLE|COST Action MitoEAGLE]] | ||
The MitoEAGLE project aims at establishing a quantitative database on mitochondrial (mt) respiratory physiology. In this context the necessity for harmonizing the terminology has become increasingly apparent. Substrate-uncoupler-inhibitor titrations (SUIT) are applied to experimentally control electron transfer | The [[MitoEAGLE]] project aims at establishing a quantitative database on mitochondrial (mt) respiratory physiology. In this context the necessity for harmonizing the terminology has become increasingly apparent. Substrate-uncoupler-inhibitor titrations ([[SUIT]]) are applied to experimentally control [[electron transfer-pathway]]s in [[mitochondrial preparations]]. Complementary to [[pathway control state]]s (PCS), [[coupling control state]]s (CCS: ET, OXPHOS, LEAK) are defined in mt-preparations, and the corresponding respiratory rates are of diagnostic significance [1]. Strategically designed SUIT protocols reveal a diversity of mt-respiratory control patterns and pathway additivity depending on species, organs, cell types, and pathophysiological states, as a hallmark of the transition from bioenergetics to mitochondrial physiology [2]. A rationale for categorizing PCS helps in selecting SUIT protocols according to the specific research question or diagnostic aim, and is essential for interpreting experimental results [3]. | ||
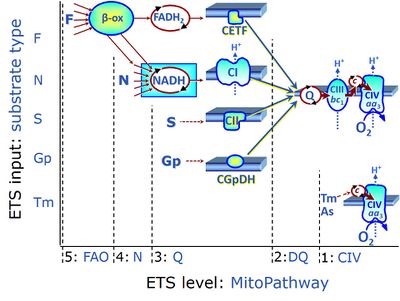
Figure 1 summarizes selected PCS, categorized according to fuel substrate types and the complexity of mitochondrial pathway types at different electron transfer- (ET-) pathway levels. ET-pathway levels are linked to ET-substrate types. The single enzyme step of Complex IV is at level 1. ET-pathway level 2 is stimulated by duroquinol (DQ) feeding electrons into Complex III (CIII) with further electron transfer to CIV and O<sub>2</sub>. ET-pathway level 3 feeds electrons from succinate to CII, and glycerophosphate (Gp) to GpDH directly upstream of the Q-junction. Electron transfer from type 4 substrates (N) feeds electrons into the N-junction from dehydrogenases and enzyme systems directly upstream of NADH and CI. The requirement of a combined operation of the F-junction and N-junction puts type F substrates to level 5 of pathway integration. F-junction substrates are fatty acids involved in β-oxidation, generating (enzyme-bound) FADH<sub>2</sub>, the substrate of electron transferring flavoprotein (CETF). In contrast, FADH<sub>2</sub> is the product of CII. A N-linked co-substrate (typically malate | Figure 1 summarizes selected PCS, categorized according to [[fuel substrate]] types and the complexity of mitochondrial pathway types at different electron transfer- (ET-) pathway levels. ET-pathway levels are linked to ET-substrate types. The single enzyme step of [[Complex IV]] is at level 1. ET-pathway level 2 is stimulated by [[duroquinol]] (DQ) feeding electrons into [[Complex III]] (CIII) with further electron transfer to [[CIV]] and O<sub>2</sub>. ET-pathway level 3 feeds electrons from [[succinate]] to [[CII]], and [[glycerophosphate]] (Gp) to [[GpDH]] directly upstream of the Q-junction. Electron transfer from type 4 substrates (N) feeds electrons into the [[N-junction]] from dehydrogenases and enzyme systems directly upstream of [[NADH]] and CI. The requirement of a combined operation of the [[F-junction]] and N-junction puts type F substrates to level 5 of pathway integration. F-junction substrates are fatty acids involved in β-oxidation, generating (enzyme-bound) FADH<sub>2</sub>, the substrate of [[electron transferring flavoprotein]] (CETF). In contrast, FADH<sub>2</sub> is the product of CII. A N-linked co-substrate (typically [[malate]] is required, and FAO can be inhibited completely by inhibition of Complex I (CI). Under physiological conditions, combinations of the fuel substrate types extend the complexity of PCS, exerting additive or competitive effects on respiratory capacity [2-5]. Analysis of combined NS- versus single N- and S-pathway capacities yields information on pathway interactions and channeling through [[supercomplex]] assemblies [4], and leads to a re-evaluation of apparent excess capacities of CIV [5]. | ||
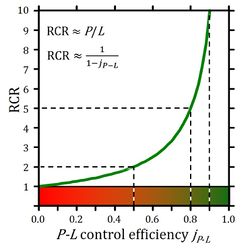
Biochemical ''cell ergometry'' aims at measurement of ''J''<sub>O2,max</sub> (compare ''V''<sub>O2,max</sub> in exercise ergometry of humans and animals) of cell respiration linked to phosphorylation of ADP to ATP. The corresponding [[OXPHOS-capacity]] is based on saturating concentrations of ADP, [ADP]*, and inorganic phosphate, [P<sub>i</sub>]*, available to the mitochondria. This is metabolically opposite to experimental uncoupling of respiration, which yields noncoupled [[ET-capacity]]. | Biochemical ''[[cell ergometry]]'' aims at measurement of ''J''<sub>O2,max</sub> (compare ''V''<sub>O2,max</sub> in exercise ergometry of humans and animals) of cell respiration linked to phosphorylation of ADP to ATP. The corresponding [[OXPHOS-capacity]] is based on saturating concentrations of ADP, [ADP]*, and inorganic phosphate, [P<sub>i</sub>]*, available to the mitochondria. This is metabolically opposite to experimental [[uncoupling]] of respiration, which yields [[noncoupled]] [[ET-capacity]]. The concept-driven terminology on CCS (LEAK, OXPHOS, ET) provides insights into the aims and rigorous quality control of diagnostic mitochondrial physiology [1]. With emphasis on conceptual differences, harmonization is achieved with the historical terminology in bioenergetics (States 1 to 5). Corresponding to the respiratory coupling states, the respiratory rates are distinguished as ''L'', ''P'', and ''E''. On a statistical basis, the classical [[respiratory acceptor control ratio]] (RCR = State 3/State 4 respiration) has to be replaced by the biochemical coupling efficiency, defined as [[OXPHOS-coupling efficiency]], ''j<sub>≈P</sub>'' = (''P-L'')/''P'' = 1-''L/P'', or [[ET-coupling efficiency]], ''j<sub>≈E</sub>'' = (''E-L'')/''E'' = 1-''L/E'', which are equivalent only at an [[Excess E-P capacity |excess ''E-P'' capacity]] equal to zero (''ExP'' = ''E-P'' = 0) (Figure 2 [4]). | ||
We | We invite scientists and students to support our effort to prepare joint publications for implementing a consistent terminology on respiratory states and rates, to ‘facilitate effective transdisciplinary communication, education, and ultimately further discovery’ and advance the quality and impact of mitochondrial physiology [1]. | ||
|editor=[[Gnaiger E]] | |editor=[[Gnaiger E]] | ||
}} | }} | ||
== Affiliations and support == | == Affiliations and support == | ||
| Line 23: | Line 23: | ||
== References == | == References == | ||
:::# | :::# Gnaiger E, Aasander Frostner E, Abdul Karim N, Abumrad NA, Acuna-Castroviejo D, Adiele RC et al (2019) Mitochondrial respiratory states and rates. MitoFit Preprint Arch doi:10.26124/mitofit:190001. - [[Gnaiger 2019 MitoFit Preprints |»Bioblast link«]] | ||
:::# Gnaiger E (2009) Capacity of oxidative phosphorylation in human skeletal muscle. New perspectives of mitochondrial physiology. Int J Biochem Cell Biol 41:1837-45. - [[Gnaiger 2009 Int J Biochem Cell Biol |»Bioblast link«]] | :::# Gnaiger E (2009) Capacity of oxidative phosphorylation in human skeletal muscle. New perspectives of mitochondrial physiology. Int J Biochem Cell Biol 41:1837-45. - [[Gnaiger 2009 Int J Biochem Cell Biol |»Bioblast link«]] | ||
:::# Doerrier C, Garcia-Souza LF, Krumschnabel G, Wohlfarter Y, Mészáros AT, Gnaiger E (2018) High-Resolution FluoRespirometry and OXPHOS protocols for human cells, permeabilized fibers from small biopsies of muscle, and isolated mitochondria. Methods Mol Biol 1782:31-70. - [[Doerrier 2018 Methods Mol Biol |»Bioblast link«]] | :::# Doerrier C, Garcia-Souza LF, Krumschnabel G, Wohlfarter Y, Mészáros AT, Gnaiger E (2018) High-Resolution FluoRespirometry and OXPHOS protocols for human cells, permeabilized fibers from small biopsies of muscle, and isolated mitochondria. Methods Mol Biol 1782:31-70. - [[Doerrier 2018 Methods Mol Biol |»Bioblast link«]] | ||
:::# Gnaiger E (2014) Mitochondrial pathways and respiratory control. An introduction to OXPHOS analysis. 4th ed. Mitochondr Physiol Network 19.12. Oroboros MiPNet Publications, Innsbruck:80 pp. - [[Gnaiger 2014 MitoPathways |»Bioblast link«]] | :::# Gnaiger E (2014) Mitochondrial pathways and respiratory control. An introduction to OXPHOS analysis. 4th ed. Mitochondr Physiol Network 19.12. Oroboros MiPNet Publications, Innsbruck:80 pp. - [[Gnaiger 2014 MitoPathways |»Bioblast link«]] | ||
:::# Lemieux H, Blier PU, Gnaiger E (2017) Remodeling pathway control of mitochondrial respiratory capacity by temperature in mouse heart: electron flow through the Q-junction in permeabilized fibers. Sci Rep 7:2840. - [[Lemieux 2017 Sci Rep |»Bioblast link«]] | :::# Lemieux H, Blier PU, Gnaiger E (2017) Remodeling pathway control of mitochondrial respiratory capacity by temperature in mouse heart: electron flow through the Q-junction in permeabilized fibers. Sci Rep 7:2840. - [[Lemieux 2017 Sci Rep |»Bioblast link«]] | ||
:::# Gnaiger E (2015) The functional design of the electron transfer system (ETS) and mitochondrial respiratory control. - [[Gnaiger 2015 Abstract MiPschool Cape Town 2015]] | |||
:::# Gnaiger E (2015) On coupling control of mitochondrial respiration. What is wrong with the RCR? - [[Gnaiger 2015 Abstract MiPschool Cape Town 2015 II]] | |||
== Figures == | |||
[[File:SUIT-catg FNSGpCIV.jpg|left|400px]] | |||
:::: '''Figure 1.''' ET-pathway control states are defined in mitochondrial preparations complementary to coupling control states (from http://www.bioblast.at/index.php/Electron_transfer-pathway_state ; see ref. 6 for further details). | |||
[[File: | [[File:RCR and OXPHOS coupling eff.jpg|right|250px]] | ||
:::: '''Figure 1.''' | :::: '''Figure 2.''' Respiratory acceptor control ratio as a function of OXPHOS-coupling efficiency, ''j<sub>P-L</sub>''. RCR is the State 3/State 4 flux ratio [4], equal to ''P''/''L'' if State 3 is at saturating [ADP] and [P<sub>i</sub>]. RCR from 1.0 to infinity is highly non-linear in the typical experimental range of RCR 3 to 10: when j''<sub>P-L</sub>'' increases from 0.8 to 0.9, RCR doubles from 5 to 10. RCR increases to infinity at the limit of ''j<sub>P-L</sub>''=1.0. Statistical analyses of RCR±SD require linearization by transformation to ''j<sub>P-L</sub>'' (from ref. 4; see ref. 7 for further details). | ||
== Questions == | |||
:::: A: Why are various substrate combinations applied in protocols for mitochondrial respiration? | |||
::::::# Can glutamate (G) be used without malate (M), and how do you interprete G-OXPHOS capacity? | |||
::::::# Can pyruvate (P) be used without M, and how do you interprete P-OXPHOS capacity? | |||
::::::# Can M be used alone, and how do you interprete M-OXPHOS capacity? | |||
::::::# Can a fatty acid (F) be used alone, and why is a low concentration of M suggested for F-OXPHOS capacity? | |||
::::::# Which role does mt-malic enzyme (mtME) play in mitochondrial pathway control using the above substrates? | |||
::::::# How do F-OXPHOS and F-ET capacities change upon titration of Rot? | |||
:::: B: Compare the significance of using succinate (S) alone or in combination with rotenone (Rot). | |||
::::::# Does S supplied alone to mitochondrial preparations establish a physiologically relevant state? | |||
::::::# Which metabolic problems occur when S is supplied alone to mitochondrial preparations, and do these problems occur generally in mt-preparations? | |||
::::::# Should S or Rot be titrated first when using intact cells or mt-preparations? | |||
:::: C: Select a SUIT protocol and interprete patterns of mitochondrial respiratory control: | |||
::::::# Which SUIT protocol(s) do you use for evaluation of a defect by dyscoupling? | |||
::::::# You suspect a specific defect of fatty acid oxidation (F-OXPHOS capacity). - Which SUIT protocol do you select? | |||
::::::# PM-ET capacity is down. How do you proof that a Complex I (CI)defect is responsible? | |||
::::::# OXPHOS capacity is down with PM as substrates (N-pathway), but PM-ET capacity is unchanged. - Where is the injury? | |||
::::::# PM-ET and S-ET capacities are similar. Adding these mathematically yields a higher value compared to the measurement of ET-capacity when these substrates are provided in combination (PMS-ET capacity). - How do you interpret such a result? | |||
::::::# PM-ET capacity is high, F-ET capacity is moderate, but addition of F after PM in the noncoupled state does not stimulate respiration. - What is the significance? | |||
:::: D: What is the difference between State 3 and OXPHOS capacity? | |||
::::::# How do you measure OXPHOS capacity? | |||
::::::# Give a definition of State 3 and OXPHOS capacity. | |||
::::::# What is the difference between a respiratory state and a respiratory rate? | |||
::::::# Why is the distinction between State 3 and OXPHOS capacity important? | |||
:::: E: Which definitions can you provide on metabolic efficiency? | |||
::::::# How are biochemical flux efficiency and thermodynamic efficiency of OXPHOS related? | |||
::::::# Is efficiency important in biological evolution? | |||
::::::# Is efficiency important in our society? | |||
{{Labeling | {{Labeling | ||
| Line 39: | Line 85: | ||
|couplingstates=LEAK, OXPHOS, ET | |couplingstates=LEAK, OXPHOS, ET | ||
|pathways=F, N, S, Gp, DQ, CIV, NS, Other combinations | |pathways=F, N, S, Gp, DQ, CIV, NS, Other combinations | ||
|event=Oral | |event=A1, Oral | ||
|additional=MitoEAGLE | |||
}} | }} | ||
Latest revision as of 23:46, 25 January 2021
| Mitochondrial states and rates: 1. Electron transfer pathways and respiratory control. 2. Coupling control. |
Link: MitoEAGLE
Gnaiger E (2018)
Event: MiPschool Tromso-Bergen 2018
The MitoEAGLE project aims at establishing a quantitative database on mitochondrial (mt) respiratory physiology. In this context the necessity for harmonizing the terminology has become increasingly apparent. Substrate-uncoupler-inhibitor titrations (SUIT) are applied to experimentally control electron transfer-pathways in mitochondrial preparations. Complementary to pathway control states (PCS), coupling control states (CCS: ET, OXPHOS, LEAK) are defined in mt-preparations, and the corresponding respiratory rates are of diagnostic significance [1]. Strategically designed SUIT protocols reveal a diversity of mt-respiratory control patterns and pathway additivity depending on species, organs, cell types, and pathophysiological states, as a hallmark of the transition from bioenergetics to mitochondrial physiology [2]. A rationale for categorizing PCS helps in selecting SUIT protocols according to the specific research question or diagnostic aim, and is essential for interpreting experimental results [3].
Figure 1 summarizes selected PCS, categorized according to fuel substrate types and the complexity of mitochondrial pathway types at different electron transfer- (ET-) pathway levels. ET-pathway levels are linked to ET-substrate types. The single enzyme step of Complex IV is at level 1. ET-pathway level 2 is stimulated by duroquinol (DQ) feeding electrons into Complex III (CIII) with further electron transfer to CIV and O2. ET-pathway level 3 feeds electrons from succinate to CII, and glycerophosphate (Gp) to GpDH directly upstream of the Q-junction. Electron transfer from type 4 substrates (N) feeds electrons into the N-junction from dehydrogenases and enzyme systems directly upstream of NADH and CI. The requirement of a combined operation of the F-junction and N-junction puts type F substrates to level 5 of pathway integration. F-junction substrates are fatty acids involved in β-oxidation, generating (enzyme-bound) FADH2, the substrate of electron transferring flavoprotein (CETF). In contrast, FADH2 is the product of CII. A N-linked co-substrate (typically malate is required, and FAO can be inhibited completely by inhibition of Complex I (CI). Under physiological conditions, combinations of the fuel substrate types extend the complexity of PCS, exerting additive or competitive effects on respiratory capacity [2-5]. Analysis of combined NS- versus single N- and S-pathway capacities yields information on pathway interactions and channeling through supercomplex assemblies [4], and leads to a re-evaluation of apparent excess capacities of CIV [5].
Biochemical cell ergometry aims at measurement of JO2,max (compare VO2,max in exercise ergometry of humans and animals) of cell respiration linked to phosphorylation of ADP to ATP. The corresponding OXPHOS-capacity is based on saturating concentrations of ADP, [ADP]*, and inorganic phosphate, [Pi]*, available to the mitochondria. This is metabolically opposite to experimental uncoupling of respiration, which yields noncoupled ET-capacity. The concept-driven terminology on CCS (LEAK, OXPHOS, ET) provides insights into the aims and rigorous quality control of diagnostic mitochondrial physiology [1]. With emphasis on conceptual differences, harmonization is achieved with the historical terminology in bioenergetics (States 1 to 5). Corresponding to the respiratory coupling states, the respiratory rates are distinguished as L, P, and E. On a statistical basis, the classical respiratory acceptor control ratio (RCR = State 3/State 4 respiration) has to be replaced by the biochemical coupling efficiency, defined as OXPHOS-coupling efficiency, j≈P = (P-L)/P = 1-L/P, or ET-coupling efficiency, j≈E = (E-L)/E = 1-L/E, which are equivalent only at an excess E-P capacity equal to zero (ExP = E-P = 0) (Figure 2 [4]).
We invite scientists and students to support our effort to prepare joint publications for implementing a consistent terminology on respiratory states and rates, to ‘facilitate effective transdisciplinary communication, education, and ultimately further discovery’ and advance the quality and impact of mitochondrial physiology [1].
• Bioblast editor: Gnaiger E
Affiliations and support
- D. Swarovski Research Lab, Dept. Visceral, Transplant Thoracic Surgery, Medical Univ Innsbruck
- Oroboros Instruments, Innsbruck, Austria
- Contribution to COST Action CA15203 MitoEAGLE, supported by COST (European Cooperation in Science and Technology), and K-Regio project MitoFit.
References
- Gnaiger E, Aasander Frostner E, Abdul Karim N, Abumrad NA, Acuna-Castroviejo D, Adiele RC et al (2019) Mitochondrial respiratory states and rates. MitoFit Preprint Arch doi:10.26124/mitofit:190001. - »Bioblast link«
- Gnaiger E (2009) Capacity of oxidative phosphorylation in human skeletal muscle. New perspectives of mitochondrial physiology. Int J Biochem Cell Biol 41:1837-45. - »Bioblast link«
- Doerrier C, Garcia-Souza LF, Krumschnabel G, Wohlfarter Y, Mészáros AT, Gnaiger E (2018) High-Resolution FluoRespirometry and OXPHOS protocols for human cells, permeabilized fibers from small biopsies of muscle, and isolated mitochondria. Methods Mol Biol 1782:31-70. - »Bioblast link«
- Gnaiger E (2014) Mitochondrial pathways and respiratory control. An introduction to OXPHOS analysis. 4th ed. Mitochondr Physiol Network 19.12. Oroboros MiPNet Publications, Innsbruck:80 pp. - »Bioblast link«
- Lemieux H, Blier PU, Gnaiger E (2017) Remodeling pathway control of mitochondrial respiratory capacity by temperature in mouse heart: electron flow through the Q-junction in permeabilized fibers. Sci Rep 7:2840. - »Bioblast link«
- Gnaiger E (2015) The functional design of the electron transfer system (ETS) and mitochondrial respiratory control. - Gnaiger 2015 Abstract MiPschool Cape Town 2015
- Gnaiger E (2015) On coupling control of mitochondrial respiration. What is wrong with the RCR? - Gnaiger 2015 Abstract MiPschool Cape Town 2015 II
Figures
- Figure 1. ET-pathway control states are defined in mitochondrial preparations complementary to coupling control states (from http://www.bioblast.at/index.php/Electron_transfer-pathway_state ; see ref. 6 for further details).
- Figure 2. Respiratory acceptor control ratio as a function of OXPHOS-coupling efficiency, jP-L. RCR is the State 3/State 4 flux ratio [4], equal to P/L if State 3 is at saturating [ADP] and [Pi]. RCR from 1.0 to infinity is highly non-linear in the typical experimental range of RCR 3 to 10: when jP-L increases from 0.8 to 0.9, RCR doubles from 5 to 10. RCR increases to infinity at the limit of jP-L=1.0. Statistical analyses of RCR±SD require linearization by transformation to jP-L (from ref. 4; see ref. 7 for further details).
Questions
- A: Why are various substrate combinations applied in protocols for mitochondrial respiration?
- Can glutamate (G) be used without malate (M), and how do you interprete G-OXPHOS capacity?
- Can pyruvate (P) be used without M, and how do you interprete P-OXPHOS capacity?
- Can M be used alone, and how do you interprete M-OXPHOS capacity?
- Can a fatty acid (F) be used alone, and why is a low concentration of M suggested for F-OXPHOS capacity?
- Which role does mt-malic enzyme (mtME) play in mitochondrial pathway control using the above substrates?
- How do F-OXPHOS and F-ET capacities change upon titration of Rot?
- A: Why are various substrate combinations applied in protocols for mitochondrial respiration?
- B: Compare the significance of using succinate (S) alone or in combination with rotenone (Rot).
- Does S supplied alone to mitochondrial preparations establish a physiologically relevant state?
- Which metabolic problems occur when S is supplied alone to mitochondrial preparations, and do these problems occur generally in mt-preparations?
- Should S or Rot be titrated first when using intact cells or mt-preparations?
- B: Compare the significance of using succinate (S) alone or in combination with rotenone (Rot).
- C: Select a SUIT protocol and interprete patterns of mitochondrial respiratory control:
- Which SUIT protocol(s) do you use for evaluation of a defect by dyscoupling?
- You suspect a specific defect of fatty acid oxidation (F-OXPHOS capacity). - Which SUIT protocol do you select?
- PM-ET capacity is down. How do you proof that a Complex I (CI)defect is responsible?
- OXPHOS capacity is down with PM as substrates (N-pathway), but PM-ET capacity is unchanged. - Where is the injury?
- PM-ET and S-ET capacities are similar. Adding these mathematically yields a higher value compared to the measurement of ET-capacity when these substrates are provided in combination (PMS-ET capacity). - How do you interpret such a result?
- PM-ET capacity is high, F-ET capacity is moderate, but addition of F after PM in the noncoupled state does not stimulate respiration. - What is the significance?
- C: Select a SUIT protocol and interprete patterns of mitochondrial respiratory control:
- D: What is the difference between State 3 and OXPHOS capacity?
- How do you measure OXPHOS capacity?
- Give a definition of State 3 and OXPHOS capacity.
- What is the difference between a respiratory state and a respiratory rate?
- Why is the distinction between State 3 and OXPHOS capacity important?
- D: What is the difference between State 3 and OXPHOS capacity?
- E: Which definitions can you provide on metabolic efficiency?
- How are biochemical flux efficiency and thermodynamic efficiency of OXPHOS related?
- Is efficiency important in biological evolution?
- Is efficiency important in our society?
- E: Which definitions can you provide on metabolic efficiency?
Labels: MiParea: Respiration, mt-Awareness
Coupling state: LEAK, OXPHOS, ET Pathway: F, N, S, Gp, DQ, CIV, NS, Other combinations
Event: A1, Oral MitoEAGLE