MiPNet06.06 Chemical O2 background: Difference between revisions
No edit summary |
Bader Helga (talk | contribs) No edit summary |
||
| Line 17: | Line 17: | ||
{{Labeling | {{Labeling | ||
|area=Instruments;methods | |area=Instruments;methods | ||
|preparations=Enzyme, Oxidase; | |preparations=Enzyme, Oxidase;biochemical oxidation | ||
|instruments=Oxygraph-2k, Protocol | |instruments=Oxygraph-2k, Protocol | ||
|additional=O2k-SOP | |additional=O2k-SOP | ||
Revision as of 15:36, 17 February 2015
| Oxygraph assay of cytochrome c oxidase activity: chemical background correction. |
» Open Access-MiPNet06.06; Versions
OROBOROS (2010-08) Mitochondr Physiol Network
Abstract: Kuznetsov AV, Gnaiger E (2010) Oxygraph assay of cytochrome c oxidase activity: chemical background correction. Mitochondr Physiol Network 06.06(07): 1-4.
Autoxidation of reduced compounds (such as ascorbate, TMPD, cytochrome c) causes a chemical background oxygen flux, which is a function of oxygen concentration and has to be subtracted fom total oxygen flux. The new DatLab software provides on-line correction for instrumental and chemical background.
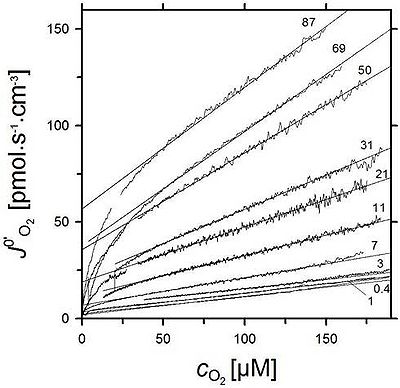
Oxygraphic determination of cytochrome c oxidase activity in the presence of TMPD, ascorbate and cytochrome c requires consideration of chemical background oxygen consumption. Several compounds are readily oxidized by molecular oxygen when dissolved in water. This leads to a significant chemical oxygen consumption in the absence of any respiring biological sample. The rate of this autoxidation represents a chemical background which strongly depends on experimental conditions, such as temperature, chemical composition, pH and oxygen concentration. Moreover, the rate of autoxidation may be significantly catalyzed by metal traces and metal-containing proteins (e.g. cytochrome c). Therefore, high-resolution respirometric measurement of oxygen flux ultimately depends on chemical background correction analyzed under experimental conditions over the entire oxygen range.
After sequential titration of ascorbate (added first) and TMPD (with or without added cytochrome c), the total oxygen flux increases due to (i) COX activity and (ii) autooxidation of ascrobate and TMPD. Correction for autooxidation is a routine procedure in high-resolution respirometry, but it requires precautions. We recommend, therefore, evaluation of chemical background effects by application of inhibitors of cytochrome c oxidase (cyanide, azide) subsequent to recording of flux with ascorbate and TMPD. After full inhibitioin of COX, a small non-COX component of biological oxidations and the large effect of autooxidation are recorded over a range of further declining oxygen concentration. After re-oxygenation, inhibited oxygen flux is recorded at oxygen levels above and within the oxygen concentration pertaining to the COX activity measurment. This provides a complete internal calibration of the chemical background, as a function of oxygen concentration.
- >> Product: OROBOROS Oxygraph-2k, O2k-Catalogue
• O2k-Network Lab: AT_Innsbruck_OROBOROS
Labels: MiParea: Instruments;methods
Preparation: Enzyme, Oxidase;biochemical oxidation
HRR: Oxygraph-2k, Protocol"Protocol" is not in the list (Oxygraph-2k, TIP2k, O2k-Fluorometer, pH, NO, TPP, Ca, O2k-Spectrophotometer, O2k-Manual, O2k-Protocol, ...) of allowed values for the "Instrument and method" property.
O2k-SOP
| Fig. 1. Continuous records of chemical background flux with 0.5 mM TMPD, 2 mM ascorbate, and various cytochrome c concentrations (μM, indicated as numbers at each curve), plotted as a function of oxygen concentration. Measured at 30 °C in mitochondrial medium R05 with 280 IU/ml catalase. The linear sections of the curves were used for background corrections at high oxygen >50 μM. |
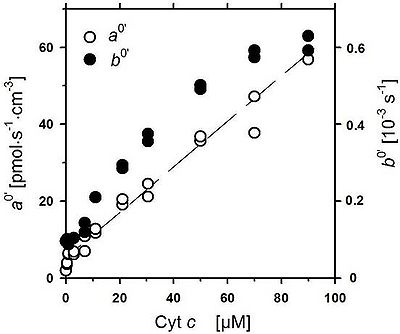
Fig. 2. Parameters of chemical background oxygen flux with TMPD+ascorbate as a function of cytochrome c concentration (Cyt c [μM). ao’ intercept and bo’ slope of linear part of oxygen dependence, from Fig.1 |
References