Gnaiger 2018 MiP2018
| Mitochondrial respiratory control by fuel substrates and specific inhibitors of respiratory enzymes: Building blocks of mitochondrial physiology Part 2. |
Link: MiP2018
Gnaiger E (2018)
Event: MiP2018
The MitoEAGLE project aims at establishing a quantitative database on mitochondrial (mt) respiratory physiology. In this context the necessity for harmonizing the terminology has become increasingly apparent. ‘Mitochondrial respiratory states and rates’ is part 1 of a series of position statements, focusing on coupling control in mt-preparations [1]. Complementary to coupling control states (CCS: ET, OXPHOS, LEAK), pathway control states (PCS) have to be defined and established experimentally. The diversity of PCS is by far higher and more complex compared to CCS, and reveals the diversity of mt-respiratory control in different species, organs, and cell types.
In this lecture I aim at providing (1) a historical account of the development of substrate-uncoupler-inhibitor-titration (SUIT) protocols to address defined PCS, as a hallmark of the transition from bioenergetics to mitochondrial physiology [2], (2) a reflection on the impact of terminological consistency for clarity of concepts, (3) a rationale for categorizing PCS in the context of comparative mitochondrial physiology, and (4) a critical evaluation of strategies for optimization of diagnostic OXPHOS protocols [3].
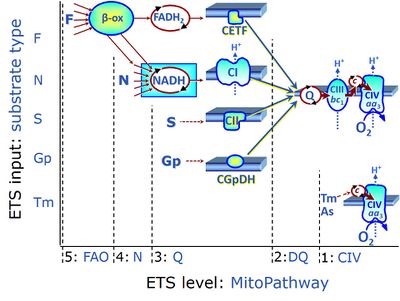
Figure 1 summarizes selected PCS, categorized according to fuel substrate types and the complexity of mitochondrial pathway types at different electron transfer- (ET-) pathway levels. ET-pathway levels are linked to ET-substrate types. The single enzyme step of Complex IV is at level 1. ET-pathway level 2 is stimulated by duroquinol (DQ) feeding electrons into Complex III (CIII) with further electron transfer to CIV and O2. ET-pathway level 3 feeds electrons from NADH, FADH2, succinate, glycerophosphate (also choline or dihydro-orotate; not shown) into respiratory complexes directly upstream of the Q-junction. Electron transfer from type 4 substrates (N) feeds electrons into the N-junction from dehydrogenases and enzyme systems directly upstream of NADH. The requirement of a combined operation of the F-junction and N-junction puts type F substrates to level 5 of pathway integration. F-junction substrates are fatty acids involved in β-oxidation, generating (enzyme-bound) FADH2, the substrate of electron transferring flavoprotein (CETF). Succinate does not belong to the type 4 substrates, since FADH2 is the product of CII, whereas FADH2Subscript text is the substrate of CETF. A N-linked co-substrate (typically malate [5]) is required, and FAO can be inhibited completely by inhibition of Complex I (CI).
Under physiological conditions, combinations of the fuel substrate types extend the complexity of PCS, exerting additive or competitive effects on respiratory capacity [2-4]. Analysis of combined NS- versus single N- and S-pathway capacities yields information on pathway interactions and channeling through supercomplex assemblies [4], and leads to a re-evaluation of apparent excess capacities of CIV [6]. The topic of mitochondrial respiratory control by fuel substrates and specific inhibitors of respiratory enzymes is suggested as part 2 in the MitoEAGLE series on ‘building blocks of mitochondrial physiology’. We cordially invite scientists to support our effort to prepare a joint publication for implementing a consistent terminology on pathway control states (PCS), to ‘facilitate effective transdisciplinary communication, education, and ultimately further discovery’ [1] and advance the quality and impact of mitochondrial physiology.
• Bioblast editor: Gnaiger E
• O2k-Network Lab: AT Innsbruck Gnaiger E
Affiliations and support
- D. Swarovski Research Lab, Dept. Visceral, Transplant Thoracic Surgery, Medical Univ Innsbruck
- Oroboros Instruments, Innsbruck, Austria
- Supported by K-Regio project MitoFit. Contribution to COST Action CA15203 MitoEAGLE.
References
- MitoEAGLE preprint 2018-09-04(41) Mitochondrial respiratory states and rates: Building blocks of mitochondrial physiology Part 1. - www.mitoeagle.org/index.php/MitoEAGLE_preprint_2018-02-08
- Gnaiger E (2009) Capacity of oxidative phosphorylation in human skeletal muscle. New perspectives of mitochondrial physiology. Int J Biochem Cell Biol 41:1837-45. - »Bioblast link«
- Doerrier C, Garcia-Souza LF, Krumschnabel G, Wohlfarter Y, Mészáros AT, Gnaiger E (2018) High-Resolution FluoRespirometry and OXPHOS protocols for human cells, permeabilized fibers from small biopsies of muscle, and isolated mitochondria. Methods Mol Biol 1782:31-70. - »Bioblast link«
- Gnaiger E (2014) Mitochondrial pathways and respiratory control. An introduction to OXPHOS analysis. 4th ed. Mitochondr Physiol Network 19.12. Oroboros MiPNet Publications, Innsbruck:80 pp. - »Bioblast link«
- Doerrier C, Garcia-Souza LF, Sumbalova Z, Krumschnabel G, Gnaiger E (2018) Evaluation of anaplerotic pathways to avoid artefacts in respirometric measurement of fatty acid oxidation. MiP2018. - »Bioblast link«
- Lemieux H, Blier PU, Gnaiger E (2017) Remodeling pathway control of mitochondrial respiratory capacity by temperature in mouse heart: electron flow through the Q-junction in permeabilized fibers. Sci Rep 7:2840. - »Bioblast link«
Figure
- Figure 1. ET-pathway control states are defined in mitochondrial preparations complementary to coupling control states. From http://www.bioblast.at/index.php/Electron_transfer-pathway_state
Labels: MiParea: Respiration, mt-Awareness
Coupling state: OXPHOS Pathway: F, N, S, Gp, DQ, CIV, NS, Other combinations
Event: Oral MitoEAGLE