Bernardo 2013 Biol Chem
| Bernardo A, De Simone R, De Nuccio C, Visentin S, Minghetti L (2013) The nuclear receptor peroxisome proliferator-activated receptor-γ promotes oligodendrocyte differentiation through mechanisms involving mitochondria and oscillatory Ca2+ waves. Biol Chem 394:1607-14. doi: 10.1515/hsz-2013-0152 |
Bernardo A, De Simone R, De Nuccio C, Visentin S, Minghetti L (2013) Biol Chem
Abstract: Peroxisome proliferator-activated receptor-γ (PPAR-γ) is one of the most studied nuclear receptor since its identification as a target to treat metabolic and neurological diseases. In addition to exerting anti-inflammatory and neuroprotective effects, PPAR-γ agonists, such as the insulin-sensitizing drug pioglitazone, promote the differentiation of oligodendrocytes (OLs), the myelin-forming cells of the central nervous system (CNS). In addition, PPAR-γ agonists increase OL mitochondrial respiratory chain activity and OL's ability to respond to environmental signals with oscillatory Ca2+ waves. Both OL maturation and oscillatory Ca2+ waves are prevented by the mitochondrial inhibitor rotenone and restored by PPAR-γ agonists, suggesting that PPAR-γ promotes myelination through mechanisms involving mitochondria.
• Bioblast editor: Gnaiger E
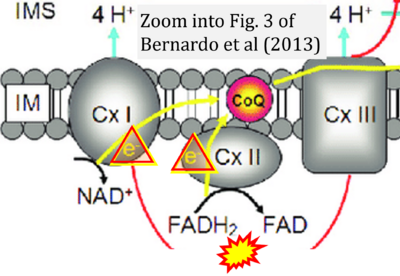
Correction: FADH2 and Complex II
- FADH2 is shown as the substrate feeding electrons into Complex II (CII). This is wrong and requires correction - for details see Gnaiger (2024).
- Gnaiger E (2024) Complex II ambiguities ― FADH2 in the electron transfer system. J Biol Chem 300:105470. https://doi.org/10.1016/j.jbc.2023.105470 - »Bioblast link«
Hydrogen ion ambiguities in the electron transfer system
Communicated by Gnaiger E (2023-10-08) last update 2023-11-10
- Electron (e-) transfer linked to hydrogen ion (hydron; H+) transfer is a fundamental concept in the field of bioenergetics, critical for understanding redox-coupled energy transformations.
- However, the current literature contains inconsistencies regarding H+ formation on the negative side of bioenergetic membranes, such as the matrix side of the mitochondrial inner membrane, when NADH is oxidized during oxidative phosphorylation (OXPHOS). Ambiguities arise when examining the oxidation of NADH by respiratory Complex I or succinate by Complex II.
- Oxidation of NADH or succinate involves a two-electron transfer of 2{H++e-} to FMN or FAD, respectively. Figures indicating a single electron e- transferred from NADH or succinate lack accuracy.
- The oxidized NAD+ is distinguished from NAD indicating nicotinamide adenine dinucleotide independent of oxidation state.
- NADH + H+ → NAD+ +2{H++e-} is the oxidation half-reaction in this H+-linked electron transfer represented as 2{H++e-} (Gnaiger 2023). Putative H+ formation shown as NADH → NAD+ + H+ conflicts with chemiosmotic coupling stoichiometries between H+ translocation across the coupling membrane and electron transfer to oxygen. Ensuring clarity in this complex field is imperative to tackle the apparent ambiguity crisis and prevent confusion, particularly in light of the increasing number of interdisciplinary publications on bioenergetics concerning diagnostic and clinical applications of OXPHOS analysis.
Labels:
Enzyme: Complex II;succinate dehydrogenase